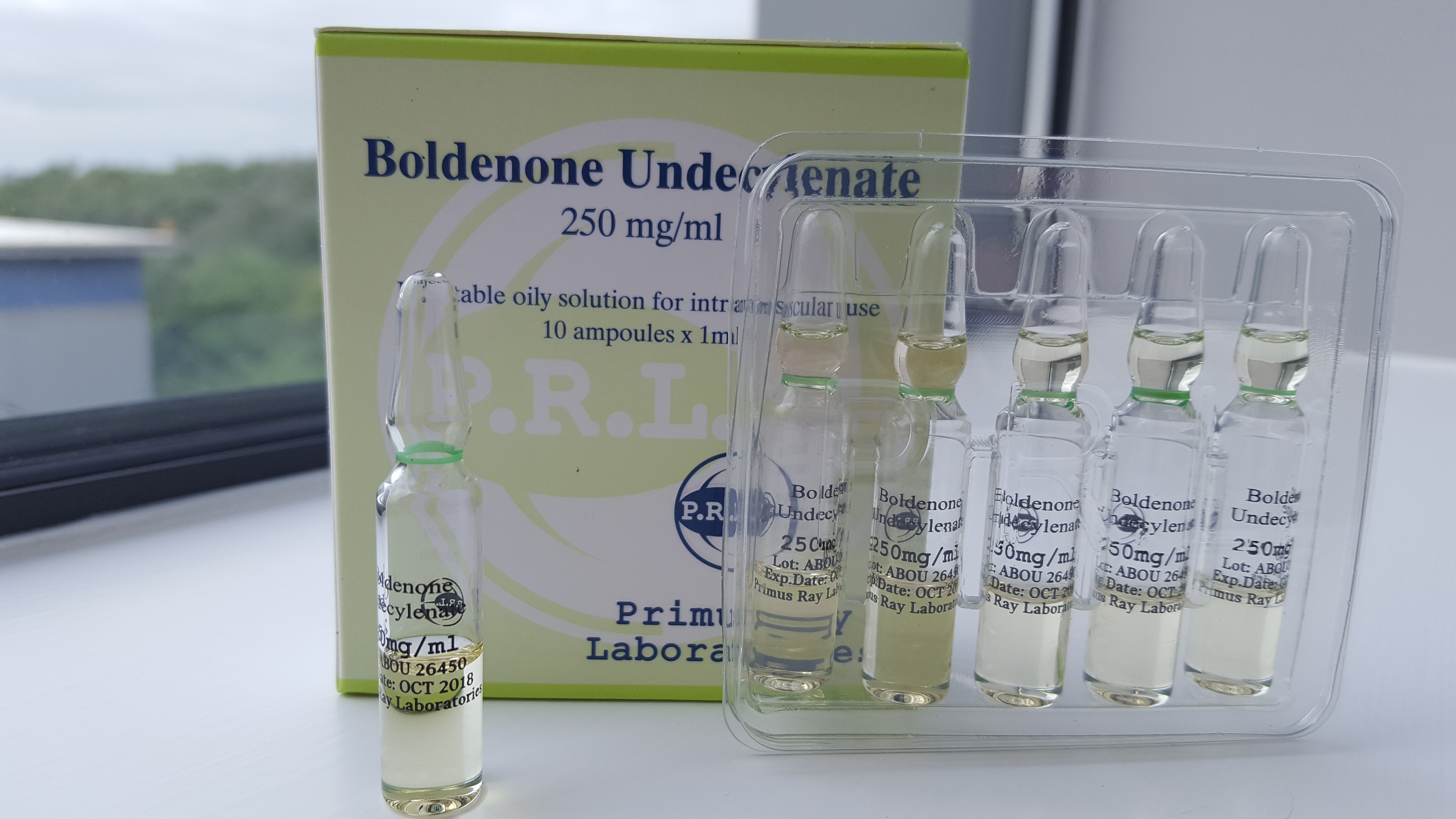
Introduction Primus Ray Laboratories Boldenone Undecylenate is labeled to contain 250 mg/ml of boldenone undecylenate and is presented in a box of ten 1-milliliter ampules. Independent testing commissioned by AnabolicLab aimed to verify the product's composition and microbiological safety. The results confirmed the product contained 241.7 mg/ml of boldenone undecylenate, a slight underdose of 3.32% […]